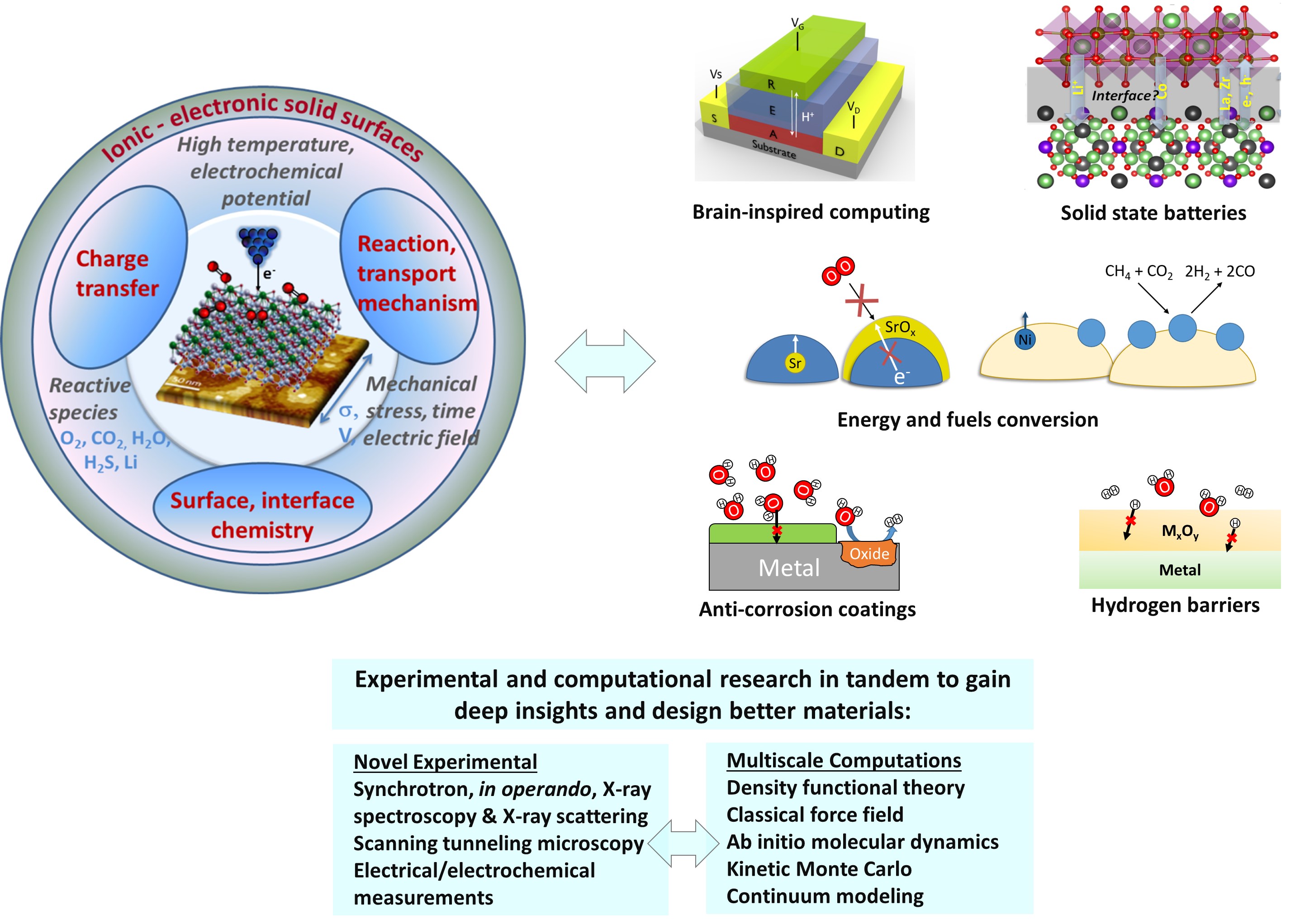
Research themes
Solid oxide fuel and electrolysis cells
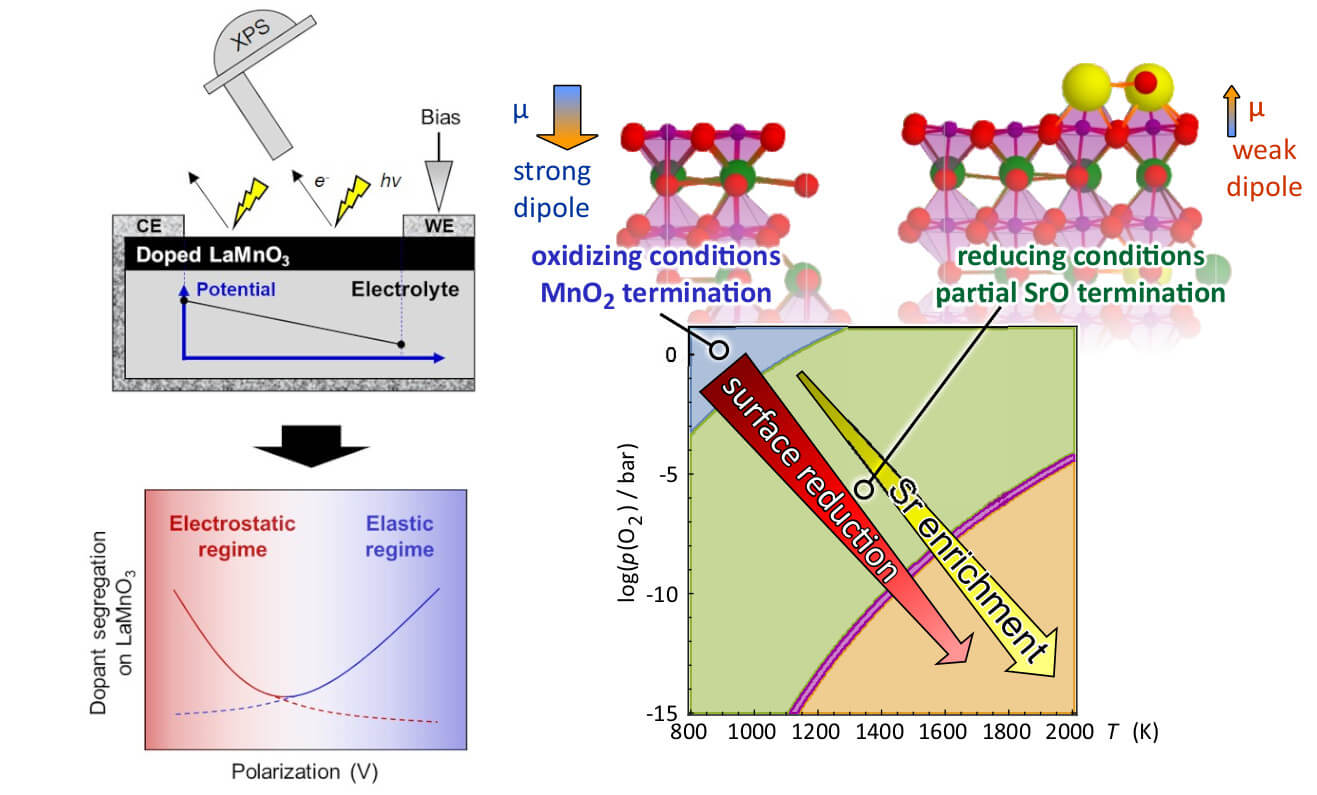
Solid oxide fuel and electrolysis cells (SOFC, SOEC) offer promising routes for clean and sustainable energy and fuels conversion routes. We investigate how cathode and anode materials (perovskite oxides) evolve at their surface, in response to electrochemical, temperature and gas stimuli in solid oxide fuel and electrolysis cells. This is important to resolve the degradation mechanisms and design better surface chemistries with enhanced durability and efficiency. We use state-of-the-art synchrotron X-ray spectroscopy techniques, and first-principles thermodynamic calculations, to resolve and predict the surface chemistry of perovskite oxides under realistic conditions.
Solid state lithium ion batteries
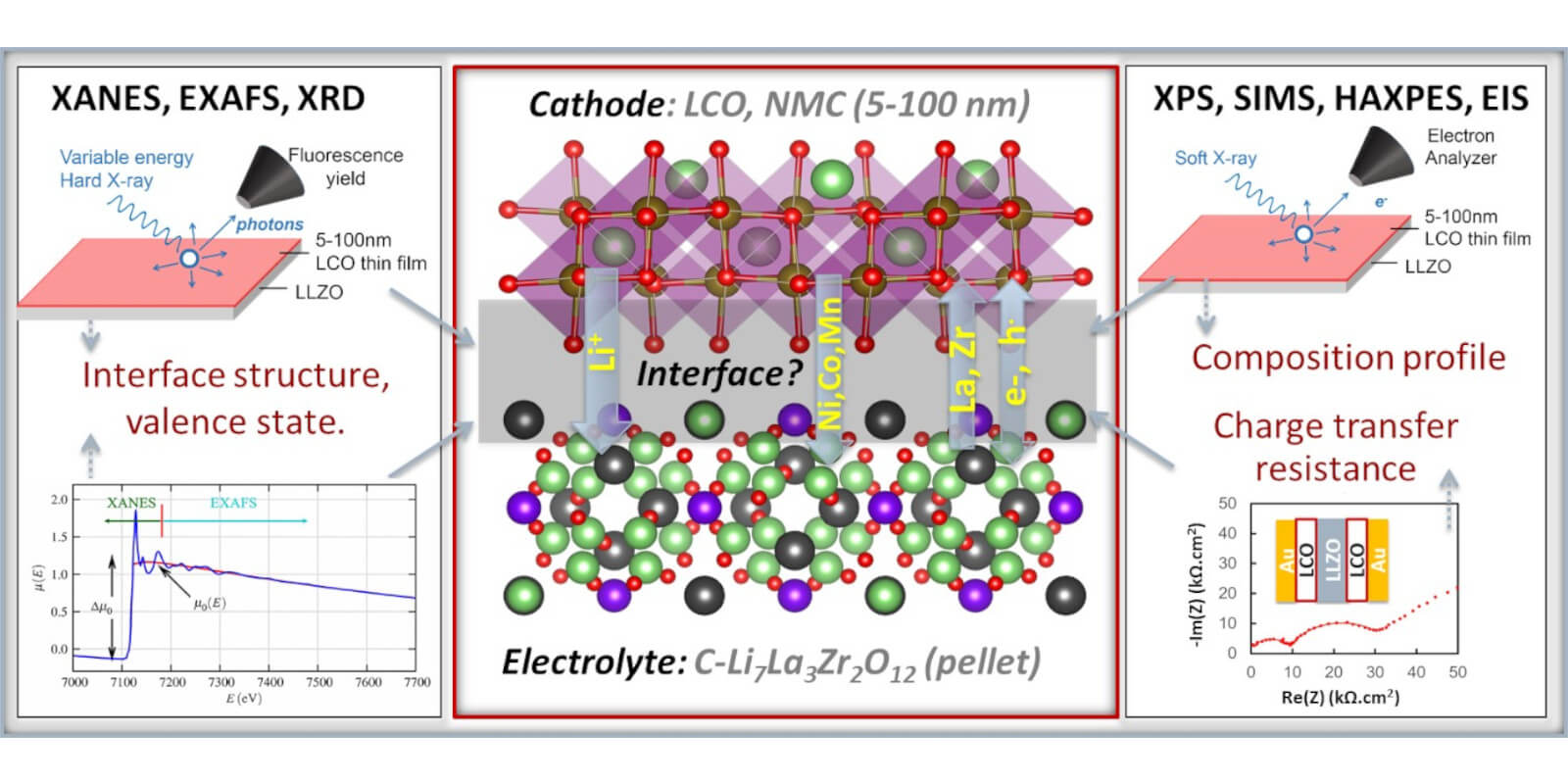
Solid state batteries with solid electrolytes offer superior energy density and safety compared to the present Li-ion batteries with liquid electrolytes. To realize their full potential in applications, we resolve the interfacial degradation in all-solid-state batteries and come up with physically-based strategies to improve their durability and charging rates. Our approach uses model systems comprising thin film cathodes on dense solid electrolyte. The design allows us to characterize cation-interdiffusion and detrimental phase formation at the buried interface and correlate those phenomena with interfacial resistance.
Hydrogen and synthetic fuel production
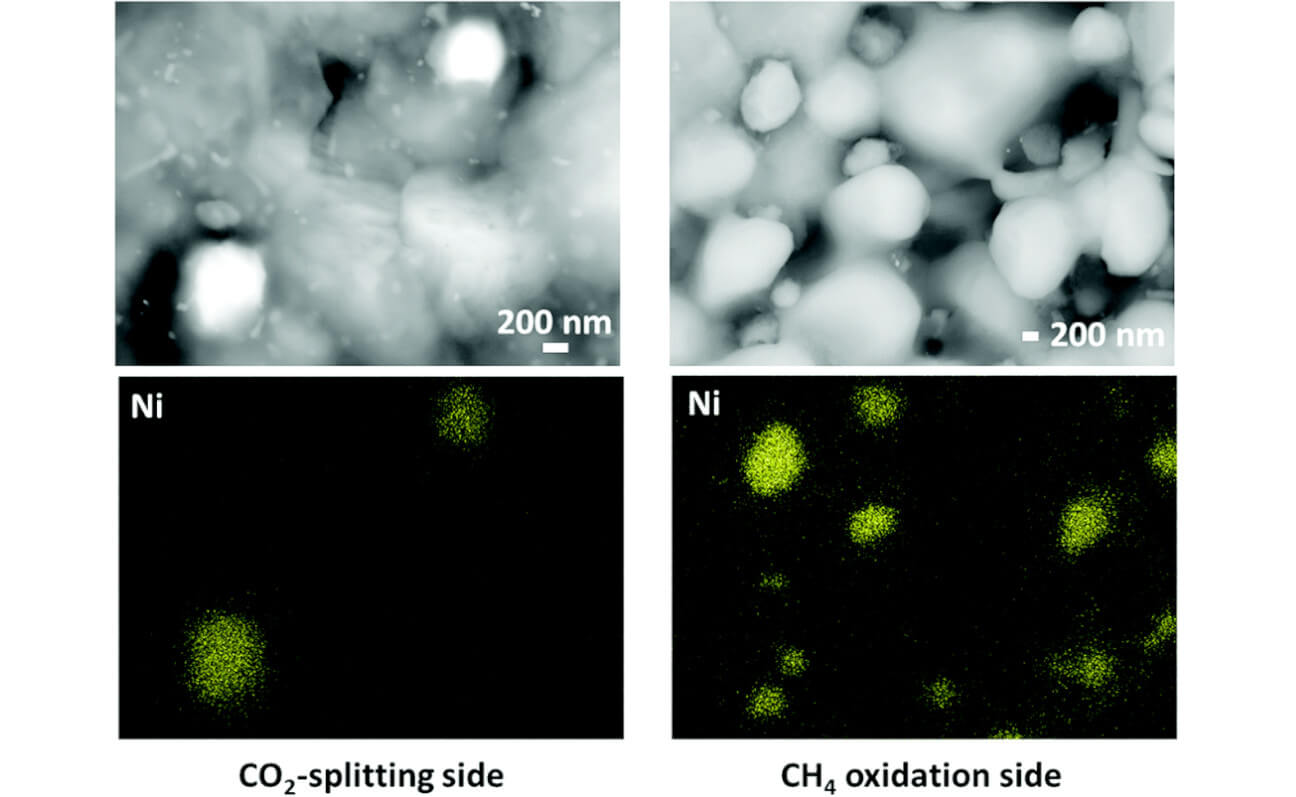
Our group also works on CO2 and H2O electrolysis in the context of establishing a sustainable energy society. Our research interests focus on in-situ probing and understanding the electrochemical interfaces during these energy conversion processes, spanning from mechanism study of model thin-film systems to the design of high-performance energy-storage materials. An ongoing project of our group is to investigate the in situ nanoparticle metal catalyst exsolution process, aiming to optimize this process to enhance the material's catalytic activity.
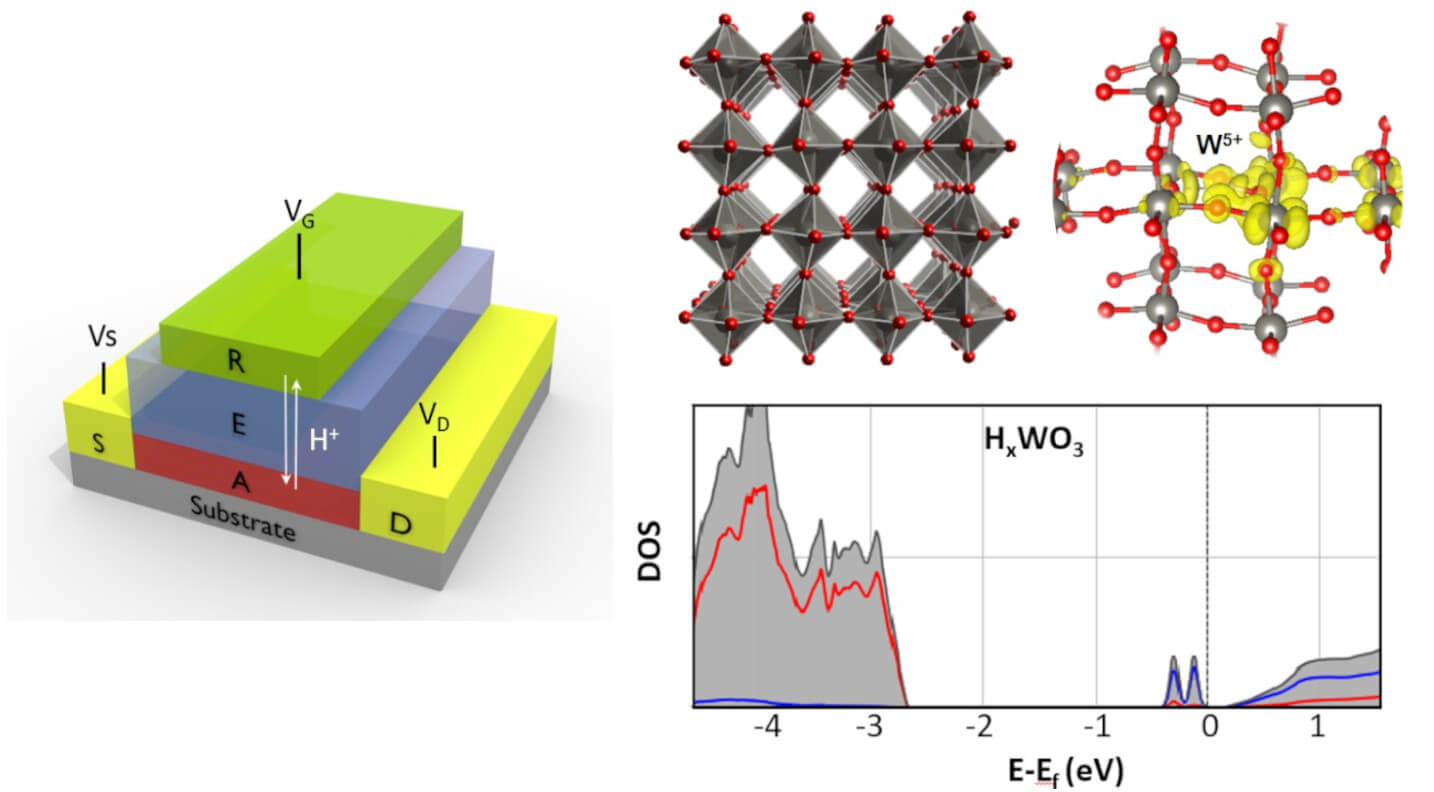
Analog brain-inspired computing
Physical neural networks made of analog resistive switching processors are promising platforms for analog computing and for emulating biological synapses. State-of-the-art resistive switches rely on either conductive filament formation or phase change. These processes suffer from poor reproducibility or high energy consumption, respectively. Our work on one hand focuses on understanding and controlling the variability of the conductive filament formation in insulating oxide materials. On the other hand, we are innovating alternative synapse designs that rely on a deterministic charge-controlled mechanism, modulated electrochemically in solid state, and that consists of shuffling the smallest cation, the proton. As typical throughout our research, here, too, we combine experimental synthesis, fabrication and characterization with first principles based computational modeling, to gain a deep understanding and control of these promising devices.
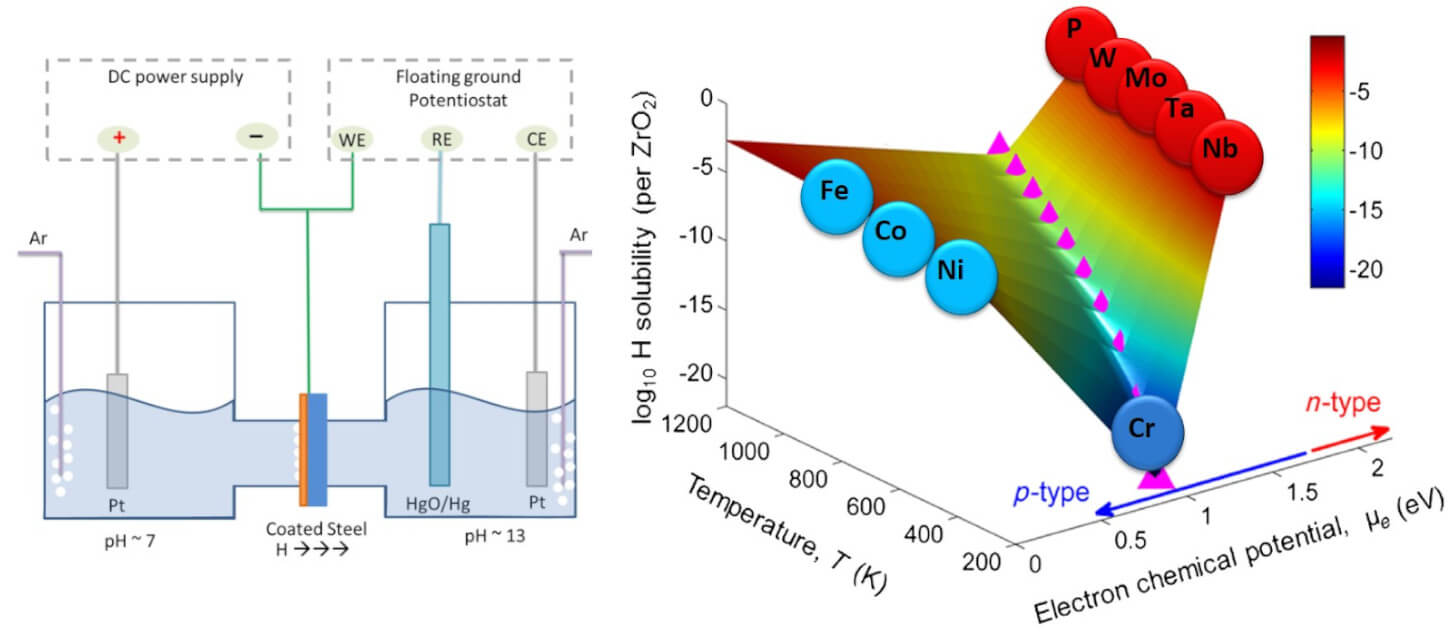
Hydrogen barrier layers
Use of hydrogen as a clean fuel will present challenges related to material durability in the infrastructure because of the embrittling nature of hydrogen when it enters metals. We develop oxide-based barrier coatings such as Al2O3, ZrO2 and SnO2 to protect infrastructure materials from hydrogen, water and other environmental stressors. By combining computational techniques such as ab initio thermodynamics, along with experiments such as permeation tests, we quantify the effect of composition and microstructure on the material’s resistance against hydrogen entry. The coatings developed can protect hydrogen storage, pumping and transportation materials, materials used in nuclear fission and fusion reactors, and steels used in geothermal well infrastructure.
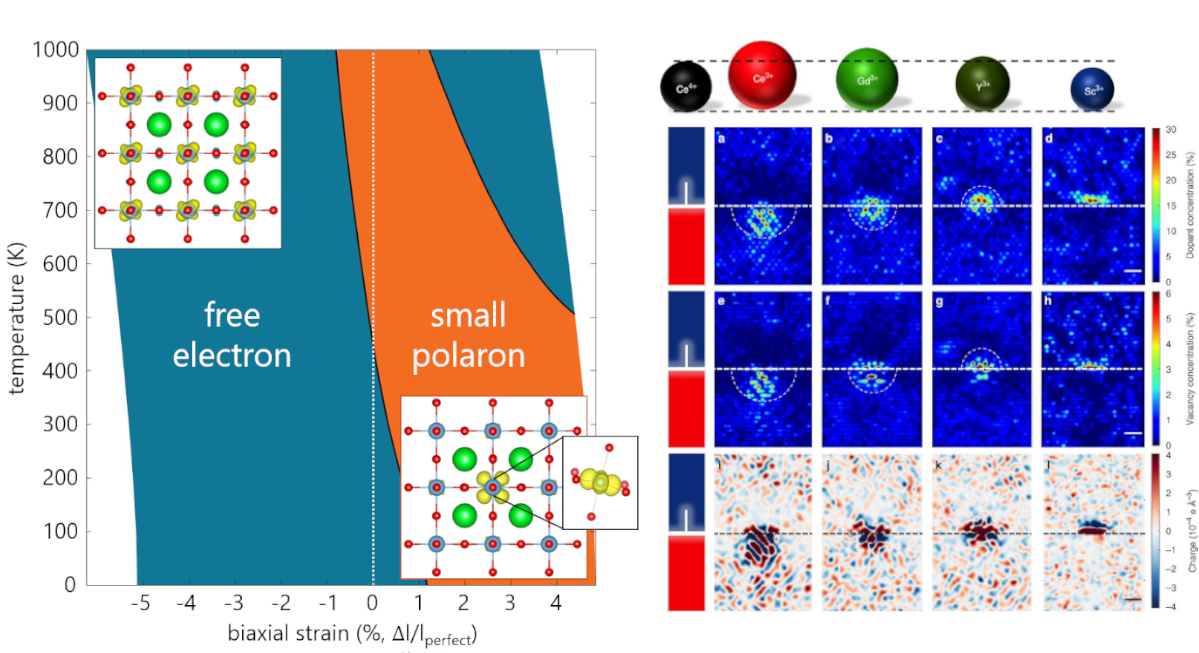
Strain and electric field effects on electronic and ionic defect transport
Strain can have a significant effect on transport properties of electronic and ionic carriers in materials. For example, elastic strain is used widely to alter the mobility of free electronic carriers in semiconductors, and it is known that strain present at cathode/electrolyte interfaces in batteries can alter the ionic conductivity of the solid Li-ion electrolyte. However, quantifying the effect of strain on charge localization and ionic conductivity is still underdeveloped. We use computational techniques such as ab initio molecular dynamics and density functional theory combined with quasiharmonic approximation calculations to assess the effect of strain and temperature on the stability and conductivity of electronic and ionic defects at finite temperatures. These findings provide guidance for conditions under which strain engineering can modulate electronic and ionic transport in electronic devices and batteries.
Facilities

We use scanning tunneling microscopy and spectroscopy (STM/STS) to determine the surface atomic and electronic structure of materials of interest. We have the unique ability to perform these measurements in reactive gases (oxygen, hydrogen, water vapor) and at elevated temperatures. We use modified Omicron VT-STM/AFM equipment which can also perform atomic force microscopy in non-contact mode.
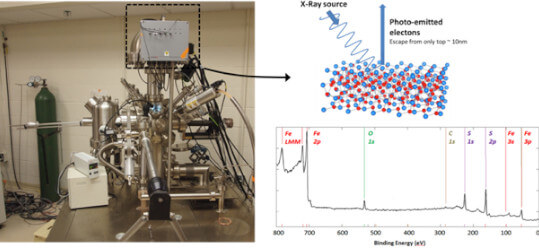
X-ray photoelectron spectroscopy (XPS), in an angle-resolved geometry, is used to analyze the top layers of a material's surface to deduce chemical information- both to detect which elements are present on the surface and their chemical binding environment and laterally-averaged electronic structure. We perform these measurements as a function of temperature, and with surfaces annealed in reactive gases in situ in our analysis chamber.

We use our computer cluster housed in MGHPCC, as well as national supercomputing infrastructures supported by NSF and DOE, to perform simulations at the electronic and atomic level, such as density functional theory, molecular dynamics and kinetic Monte Carlo simulations. (Images above taken from the TACC, NERSC and MGHPCC websites.)
